Cassiterite, chemically identified as tin dioxide (SnO₂), is the primary ore and most significant natural source of metallic tin. Belonging to the rutile mineral group, it typically crystallizes in the tetragonal system, frequently manifesting as short prismatic or bipyramidal crystals, often characterized by distinctive “elbow” twinning. While its coloration is predominantly dark brown to black due to iron impurities, it can also appear in reddish-brown, yellow, or even colorless transparent forms in rare, high-purity specimens. One of its most defining physical attributes is its high specific gravity, ranging from 6.8 to 7.1, which is exceptional for a non-metallic mineral. This density, combined with an adamantine to sub-metallic luster and a Mohs hardness of 6 to 7, makes it both chemically stable and resistant to physical weathering. Specialized varieties include “Wood Tin,” a botryoidal or fibrous form with concentric banding reminiscent of wood grain, and “Stream Tin,” referring to the rounded, water-worn pebbles found in secondary deposits.

The formation of cassiterite is genetically linked to evolved granitic magmatism and high-temperature hydrothermal processes. It typically precipitates in the late stages of magmatic differentiation, where tin-bearing volatile fluids become concentrated in pegmatites and greisens. These fluids, often enriched in boron, fluorine, and phosphorus, circulate through fractures in the host rock, depositing cassiterite alongside minerals such as quartz, wolframite, tourmaline, and fluorite. In addition to these primary vein deposits, cassiterite occurs in contact metamorphic environments, such as skarns, where magmatic intrusions interact with carbonate rocks. Due to its extreme resistance to chemical decomposition and its high density, cassiterite is frequently liberated from its primary matrix through erosion. It then undergoes mechanical concentration by fluvial or marine action to form economically vital alluvial or placer deposits, which historically provided the bulk of the world’s tin production.
Cassiterite is synonymous with the technological transition of human civilization into the Bronze Age. As early as 3000 BCE, ancient metallurgical practices involved the reduction of cassiterite in charcoal fires to produce tin, which was then alloyed with copper to create bronze—a material significantly harder and more castable than pure copper. This innovation revolutionized the production of tools, weaponry, and ceremonial objects. The strategic importance of tin ore led to the establishment of extensive ancient trade routes, with seafaring cultures like the Phoenicians venturing as far as the “Cassiterides” (traditionally associated with Cornwall and the Scilly Isles) to secure supplies. During the Industrial Revolution, the demand for cassiterite surged, driving advancements in deep-shaft mining and steam-pump technology. In a modern context, while its historical role in metallurgy remains, cassiterite is essential for the global electronics industry, serving as the raw material for the lead-free solders required in semiconductor and circuit board manufacturing.
Crystal Structure and Physico-Chemical Properties of Cassiterite
Cassiterite crystallizes in the tetragonal crystal system, specifically within the P4₂/mnm space group, a structure it shares with other members of the rutile group. The atomic arrangement consists of tin (Sn) cations coordinated with six oxygen (O) anions in an octahedral geometry, while each oxygen atom is surrounded by three tin atoms in a planar trigonal configuration. This tightly packed lattice contributes to the mineral’s remarkable density and high refractive index. Morphologically, cassiterite often develops as short, stout prismatic or dipyramidal crystals. A hallmark of its crystal habit is the frequent occurrence of contact or penetration twinning, particularly the “elbow” twin, which forms a characteristic 112° angle. This structural robustness is reflected in its physical durability, manifesting as a Mohs hardness of 6 to 7 and a lack of distinct cleavage, typically resulting in a subconchoidal to uneven fracture.
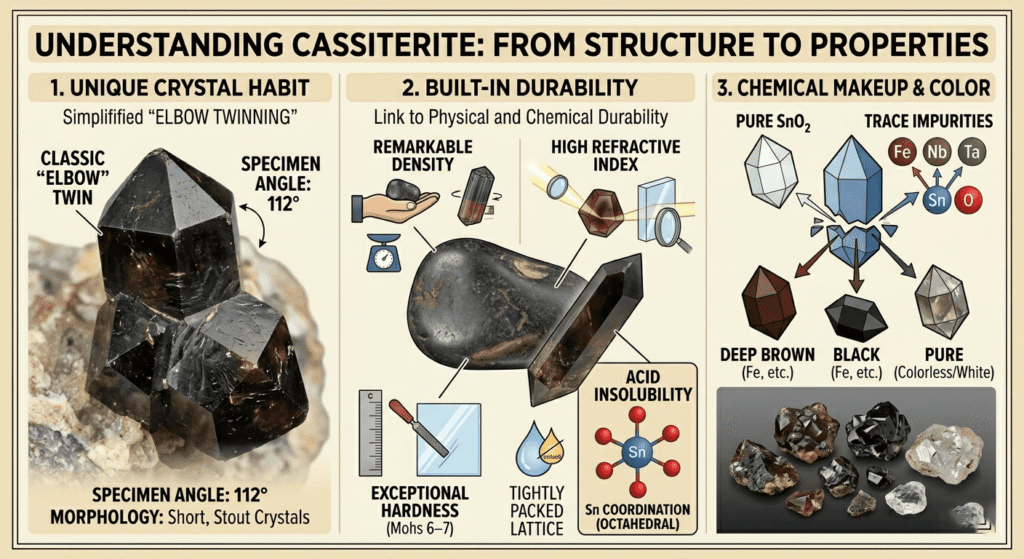
Chemically, cassiterite is primarily composed of tin dioxide (SnO₂), though natural specimens rarely exist in a pure state. The crystal lattice often incorporates minor amounts of iron, niobium, tantalum, manganese, and tungsten through ionic substitution. These trace elements are responsible for the mineral’s broad color spectrum, as pure SnO₂ is naturally colorless or white; iron impurities, in particular, induce the common deep brown to black pigmentation. One of the most significant chemical properties of cassiterite is its extreme refractory nature and relative insolubility in acids, which allows it to persist in the environment long after its host rock has eroded. Furthermore, its high specific gravity of 6.8 to 7.1 serves as a critical diagnostic property, distinguishing it from visually similar but lighter minerals like tourmaline or sphalerite. This combination of structural integrity and chemical stability ensures that cassiterite remains the most reliable and concentrated source of tin for industrial extraction.
Comprehensive Varieties of Cassiterite
Cassiterite is categorized into several distinct varieties based on its physical form, growth environment, and chemical composition. While the mineral is fundamentally tin dioxide (SnO₂), these variations reflect different geological histories and have served various industrial and decorative purposes throughout history.
Primary and Industrial Varieties
Tin Stone
This is the general term used by miners for the common, massive, or crystalline form of cassiterite found in its original host rock. It is the primary industrial ore of tin and typically occurs in high-temperature hydrothermal veins.

Stream Tin
These are rounded, water-worn pebbles or coarse sand found in alluvial or placer deposits. Due to its high specific gravity (6.8–7.1) and chemical stability, cassiterite resists weathering and concentrates in riverbeds, making this the most commercially accessible variety historically.

Float Tin
This refers to fragments of cassiterite that have broken off from a primary vein and are found scattered on hillsides or in soil. Geologists often use float tin as a “trail” to locate buried primary ore deposits.
Black Tin
A metallurgical term for cassiterite ore that has been dressed (concentrated) and is ready for smelting.

Morphological and Textural Varieties
Wood Tin
A botryoidal (grape-like) or reniform (kidney-shaped) variety formed at lower temperatures. It features a radiating fibrous structure with concentric banding of different colors, closely resembling tree rings.

Toad’s Eye Tin
A rare and specific subtype of wood tin characterized by very small, spherical, or semi-spherical banded structures. The name comes from the visual similarity of these tiny clusters to the eye of a toad.

Needle Tin
This variety consists of very thin, elongated, needle-like or hair-like crystals, often found in specialized hydrothermal environments.

Sparable Tin
An old Cornish mining term for small, sharp, diamond-shaped crystals that resemble “sparables”—the short, square-headed nails used in traditional cobbling.

Dough Tin
An extremely rare, soft, or clay-like variety that represents a poorly crystallized or amorphous state of tin oxide.

Visual and Gem-Quality Varieties
Gem-Quality Cassiterite
These are rare, transparent to translucent crystals with exceptional brilliance. While most cassiterite is opaque and dark, gem-grade specimens can be golden-yellow, wine-red, or even colorless.

Colloform Cassiterite: A broad morphological category for cassiterite that grows in rounded, globular, or grape-like masses, encompassing both wood tin and toad’s eye tin.
Chemical and Compositional Varieties
Niobian / Tantalian Cassiterite
Varieties containing trace amounts of niobium (Nb) or tantalum (Ta). These are of high interest to geochemists for studying the evolution of rare-metal magmas.

Ainalite
A variety containing a high percentage (up to 10%) of tantalum pentoxide (Ta₂O₅). It is usually found in complex pegmatites where tantalum substitutes for tin in the crystal lattice.

Ferrian Cassiterite: The most common variety, which is rich in iron (Fe). The iron content is what typically gives cassiterite its characteristic deep brown to opaque black color.
Applications and Metaphysical Significance of Cassiterite
Cassiterite serves as the indispensable backbone for global manufacturing as the primary source of tin. Beyond the production of bronze alloys and corrosion-resistant coatings for food containers, tin dioxide derived from cassiterite is utilized in the creation of transparent conductive oxides. These are essential for the functionality of modern touchscreens, liquid crystal displays, and energy-efficient window coatings. In the world of fine jewelry, rare gem-quality cassiterite, prized for its intense brilliance and high refractive index, is faceted into exquisite collectors’ stones. Additionally, the mineral’s high opacity makes it a traditional opacifying agent in the ceramic industry for creating white glazes and specialized enamels.

Beyond its physical utility, cassiterite holds profound symbolic and spiritual meaning within various lithotherapy traditions. Frequently referred to as a stone of transition, it is believed to provide the grounding necessary to navigate major life changes. Due to its remarkable density and high specific gravity, practitioners associate cassiterite with the root chakra, viewing it as a stabilizing force that anchors the user during periods of chaos. Its historical role in bridging the Stone Age to the Bronze Age lends it a reputation as a catalyst for human progress and the manifestation of intellectual ideas into physical reality. In meditative practices, the mineral is used to balance the intellect with emotion, helping to clear mental clutter and sharpen focus. Finally, its chemical stability and refractory nature are often seen as symbolic of inner strength and the ability to withstand external pressures without degradation.
