Orthoclase is a very common mineral that makes up a large part of the Earth’s crust. It belongs to the feldspar family, specifically the potassium feldspars, and has the chemical formula KAlSi3O8. You can find it in many everyday rocks like granite, where it often appears as pink, white, or grey crystals. One of its most famous features is how it breaks: it has two flat “cleavage” planes that meet at a perfect 90-degree angle. This is actually where its name comes from, as “orthos” means “straight” and “klasis” means “fracture” in Greek. It is also a very durable mineral, used as the official standard for level 6 on the Mohs scale of hardness.

Orthoclase mainly forms from the cooling of molten rock, or magma. When magma rich in potassium and silica cools down slowly deep underground, orthoclase crystals begin to grow. This is why it is so common in “plutonic” rocks like granite. If the magma cools extremely fast (like during a volcanic eruption), a different version called sanidine forms instead. Orthoclase can also be created during high-pressure metamorphism, where existing rocks are heated and squeezed until their minerals reorganize. Over millions of years, however, if orthoclase is exposed to water and weather at the surface, it eventually breaks down into soft clay minerals like kaolinite.In terms of scientific history, orthoclase was officially named in 1823 by the German mineralogist August Breithaupt. For centuries, humans have used it for practical purposes; because it helps other materials melt more easily, it is a key ingredient in making glass and ceramics. It also has a special place in geology because it contains a radioactive version of potassium. Geologists measure how this potassium decays into argon gas to calculate the exact age of rocks that are millions of years old. Finally, while most orthoclase is used for industry, clear or shimmering varieties—known as Moonstone—have been used in jewelry for thousands of years due to their unique, glowing appearance.

Optical Properties and Microscopic Features of Orthoclase
 |
This whitish orthoclase crystal exhibits low birefringence color under crossed polars. |
 |
Orthoclase crystal showing extinction patterns. |
 |
Notice the cleavage of 90 degrees in the center of the crystal. |
 |
Orthoclase crystals colorless under normal polarized light, very low birefringence colors (grey or white) under cross-polarized light. |
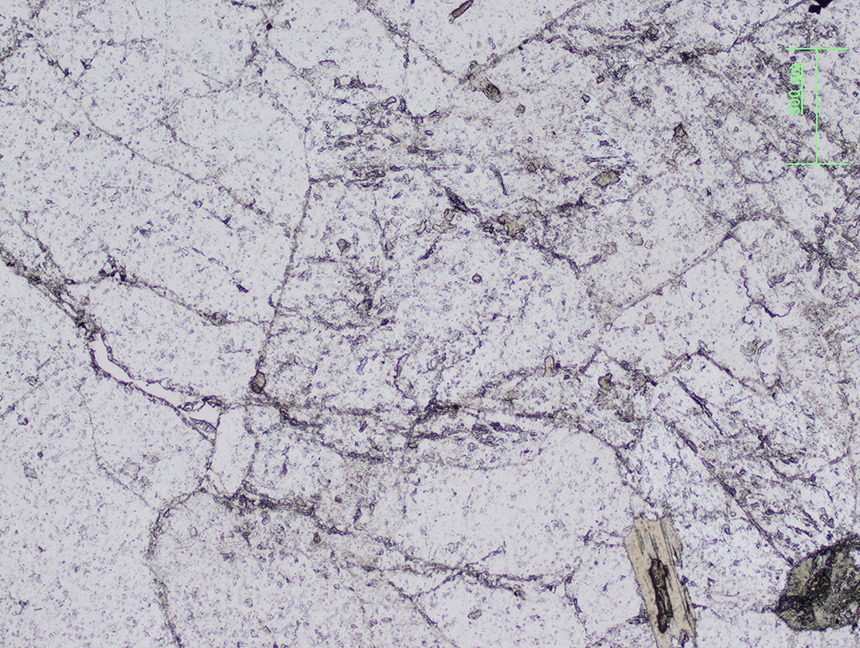 |
Orthoclase crystal surrounding plagioclase crystal. |
What is Orthoclase actually used for?
Orthoclase plays a dual role as both a critical industrial raw material and a vital tool in geological research. Its primary commercial application is in the ceramics and glass industries, where it functions as a fluxing agent. Because orthoclase contains a high concentration of potassium, it effectively lowers the melting temperature of silica during the firing process. This reduces energy consumption and results in more durable finished products, such as porcelain, sanitaryware, and wall tiles. In the glass sector, it is used to improve the chemical durability and hardness of glass containers and fiberglass. Additionally, due to its consistent hardness of 6 on the Mohs scale, finely ground orthoclase is often employed as a mild abrasive in scouring powders and polishing agents.

In the field of geosciences, orthoclase is fundamentally important for its role in geochronology. Because the mineral naturally contains the radioactive isotope Potassium-40, it acts as a geological clock. Scientists utilize Potassium-Argon and Argon-Argon dating methods to measure the decay of potassium into argon gas trapped within the crystal lattice. This process allows researchers to determine the absolute age of rock formations, helping to map the timeline of volcanic eruptions and tectonic movements throughout Earth’s history. While most orthoclase is consumed by heavy industry, high-clarity or visually unique varieties are also utilized in the gemological sector. The most notable example is Moonstone, which is valued for its optical interference patterns and is precision-cut for use in jewelry.
