Ekanite is one of the rarest and most scientifically significant gemstone minerals documented in gemology. Unlike traditional gemstones valued for their optical brilliance and physical durability, ekanite is distinguished by its specific chemical composition and inherent radioactivity. The mineral was first discovered in 1953 within the alluvial gem gravels of Sri Lanka and was subsequently named in honor of mineralogist F. L. D. Ekanayake, who first identified the specimen. As a calcium thorium silicate, ekanite contains radioactive isotopes of thorium and often uranium, which subject the mineral to a process known as metamictization. During this process, the internal crystal lattice is gradually disrupted by radioactive decay, eventually transforming the material into an amorphous or glass-like state. This characteristic makes ekanite a subject of study not only for gem collectors but also for researchers interested in the long-term effects of radiation on crystalline structures.


Formation and Geological Origin of Ekanite
The formation of ekanite is primarily associated with high-temperature, contact metamorphic environments and specific types of igneous activity. It typically occurs in areas where silica-rich fluids interact with limestone or other calcium-rich rocks under intense heat and pressure. This process often takes place in contact zones known as skarns, where the introduction of rare-earth elements and radioactive isotopes like thorium and uranium from intruding magma allows for the crystallization of calcium thorium silicate.
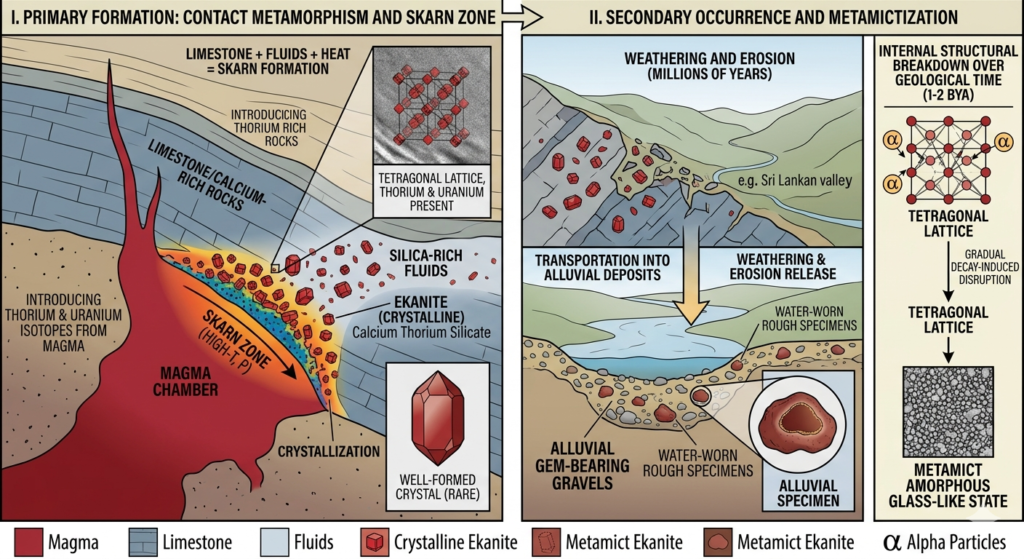
In its primary geological setting, ekanite crystallizes as a tetragonal mineral. However, its most famous occurrence is in the secondary alluvial deposits of Sri Lanka. In these locations, the mineral has been weathered out of its original host rock over millions of years and transported by water into gem-bearing gravels. Over geological timescales, the radioactive decay of thorium and uranium within the mineral’s own structure leads to its gradual transition from a crystalline state to a metamict or amorphous state. This unique evolutionary path—from high-temperature metamorphic crystallization to internal structural breakdown—makes ekanite a significant subject for geochronological and mineralogical research.
Color and Appearance
Ekanite exhibits a specific range of visual characteristics, primarily manifesting in various shades of green, such as yellowish-green, olive-green, and brownish-green. Less frequent occurrences include specimens that appear gray or nearly colorless. In its natural state, the mineral typically displays a vitreous luster and its transparency varies from translucent to opaque. Due to the internal structural damage caused by prolonged radioactive decay, well-defined crystals are exceptionally rare. This structural degradation often results in a more massive or water-worn appearance in rough specimens, which significantly increases the value of high-quality or intact crystals for both gemological collectors and scientific researchers.

Radioactivity and Safety Profile
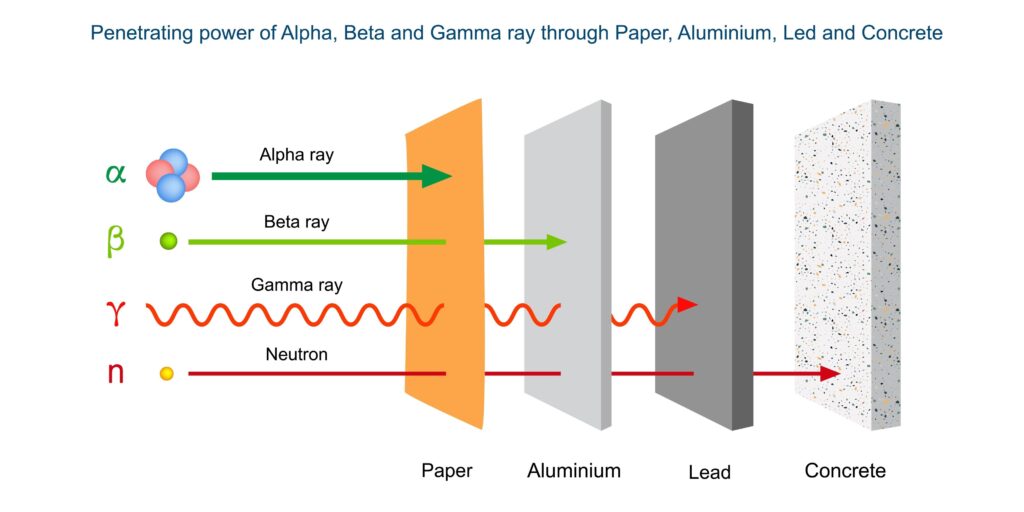
The defining scientific characteristic of ekanite is its inherent radioactivity. As a calcium thorium silicate, the mineral contains significant concentrations of thorium (Th), and frequently uranium (U), as part of its essential chemical structure. The radioactive decay of these elements emits alpha, beta, and gamma radiation, the intensity of which depends on the specific concentration of the isotopes within a given specimen.
Over geological time, this internal radiation causes the phenomenon of metamictization. The alpha particles emitted during decay collide with the mineral’s crystal lattice, systematically displacing atoms from their original positions. This process eventually collapses the orderly tetragonal structure, transforming the ekanite into an amorphous, glass-like state. While this makes the mineral a fascinatng subject for geochronological study, it also dictates specific handling and storage protocols for collectors.

From a safety perspective, while a single small ekanite gemstone generally does not pose an immediate acute health risk if handled briefly, it should be managed with caution. The primary concern is the cumulative exposure to gamma radiation and the potential inhalation of radon or thoron gas—radioactive byproducts of the decay chain—if the specimen is kept in an unventilated space. Collectors are advised to store ekanite specimens in lead-lined containers or well-ventilated areas away from living quarters. Furthermore, ekanite should never be worn as jewelry in direct contact with the skin for extended periods, and any dust generated from damaged or rough specimens should be treated as a hazardous bio-contaminant.
Ekanite serves as a significant case study in mineralogy, illustrating the complex intersection between crystalline order and radioactive decay. As a thorium-bearing silicate, it is defined not only by its rare olive-green coloration but also by the process of metamictization, where internal radiation gradually transitions the mineral from a structured lattice into an amorphous state. This unique characteristic provides researchers with a natural laboratory to observe the long-term effects of radioactive isotopes on solid matter over millions of years.From its initial discovery in the gem gravels of Sri Lanka to its classification as a highly specialized collector’s mineral, ekanite remains a subject of distinct scientific interest. Its dual nature—as both a geological product of contact metamorphism and a victim of its own internal chemical instability—places it in a unique category of “living” minerals. For the scientific community and advanced collectors, the value of ekanite lies in this transformative history. Maintaining its integrity through proper storage and handling remains a fundamental requirement for the ongoing study and preservation of this rare calcium thorium silicate.
