Cuprite is a copper(I) oxide mineral and a significant secondary mineral found within the oxidation zones of copper ore deposits. It typically forms through the chemical weathering of primary copper sulfides, such as chalcopyrite, when exposed to oxygen-rich meteoric environments over extended geological periods. This paragenetic process often results in the crystallization of cuprite alongside other secondary minerals like malachite, azurite, and native copper. These crystals frequently exhibit isometric habits, including cubic or octahedral forms, as recorded in the: Handbook of Mineralogy ,these crystals frequently exhibit isometric habits, including cubic or octahedral forms.
The nomenclature of the species is derived from the Latin word cuprum, which directly translates to “copper,” reflecting both its high metallic content and its primary chemical composition. While it serves as a minor ore of copper, its gemological significance lies in its exceptional refractive index of 2.849, which surpasses that of diamond. However, due to its relatively low Mohs hardness of 3.5 to 4.0, cuprite is primarily valued as a collector’s gemstone rather than for use in traditional jewelry. Its deep red color and adamantine luster remain its most defining diagnostic features for identification in the field, as documented in the Mindat.org mineral database.

Optical Properties and Visual Characteristics
Cuprite is characterized by its exceptionally saturated color profile, primarily manifesting in deep, coppery red to brownish-red hues. This coloration is a direct result of its high copper content and its unique crystal chemistry, which allows for a range of appearances from a translucent, vivid crimson—often compared to fine ruby—to a nearly opaque, metallic black in larger, more massive specimens. When viewed in thin fragments or under strong transmitted light, even the darkest crystals typically reveal a signature internal fire of intense red.
The optical appeal of this species is further enhanced by its adamantine to sub-metallic luster, a consequence of its high refractive index. When faceted, gem-quality cuprite displays a brilliance that can surpass that of a diamond, though such transparent material is exceedingly rare. In addition to the standard crystalline form, cuprite occasionally occurs in a capillary variety known as chalcotrichite, which consists of matted, needle-like crystals that exhibit a distinct silky appearance.

Durability and Lapidary Use
In gemological practice, cuprite is significantly more prevalent in mineral collections than in commercial jewelry due to its inherent physical limitations. With a Mohs hardness rating of 3.5 to 4.0, the species is highly susceptible to surface scratching and abrasion, making it largely unsuitable for high-impact applications such as ring stones. While pendants, earrings, and brooches represent lower-risk alternatives, any jewelry application requires protective settings to maintain the integrity of the stone.

Most cuprite crystals are characterized by small dimensions or high opacity, which limits their suitability for traditional faceting. However, cuprite frequently forms as a constituent of multi-mineral aggregates alongside other secondary copper minerals like malachite, chrysocolla, and azurite. These mixed mineral specimens are highly valued by lapidaries, who typically process them into cabochons to showcase the distinctive color contrasts and patterns formed by the various copper-bearing species.
Identification and Authentication
To identify cuprite, gemologists analyze its extreme physical and optical constants, which distinguish it from more common red stones like garnet, ruby, or spinel. The most definitive indicator is its extraordinary refractive index of 2.848—significantly higher than a diamond’s—which creates a subtle, bluish metallic luster under reflected light, while transmitted light reveals its signature deep crimson body color. Furthermore, cuprite is exceptionally dense, with a specific gravity between 6.0 and 6.14. This high density helps separate it from other “over-the-limit” (OTL) red minerals like proustite or wulfenite. As an isometric mineral, it is naturally singly refractive, though anomalous birefringence may occur due to surface stress from traditional diamond-paste polishing; professional studies suggest using alkaline silica solutions to maintain its true optical integrity.While cuprite has been synthesized for industrial research and archaeological restoration, lab-grown material is not widely used in the gemstone market. Historical simulants include devitrified glass “Maple Stones” or glass beads colored with copper(I) oxide, yet natural cuprite remains the standard for collectors. A brownish-red streak is diagnostic but should never be attempted on faceted stones as it is a destructive test. Because cuprite generally receives no treatments or enhancements, its identification remains focused on confirming its natural oxide chemistry and remarkable light-handling properties.

Is it Cuprite or Red Garnet?
While cuprite and red garnets can look nearly identical at first glance, they are actually very different minerals. The most practical way to tell them apart is by their weight and luster. Cuprite is much denser than garnet; if you hold a piece of each in your hands, the cuprite will feel significantly heavier for its size. Additionally, cuprite has a brilliant metallic or “greasy” shine, whereas garnets typically have a glassy luster similar to window glass.
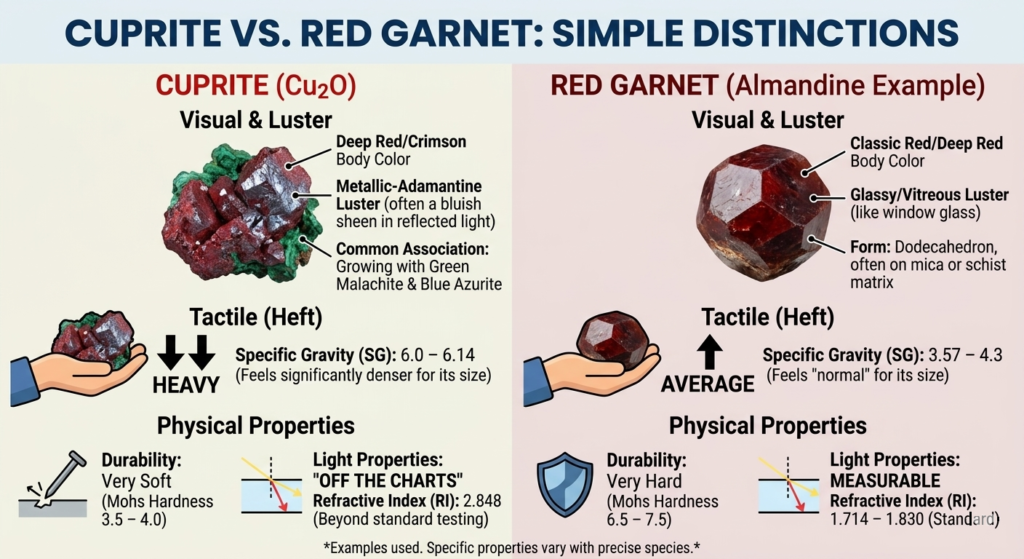
Another key difference is durability. Garnets are hard and tough, making them perfect for everyday jewelry like rings. Cuprite, however, is quite soft and can be easily scratched by a steel nail or even a common copper coin. If you look at the crystals in their natural state, cuprite is often found growing alongside green malachite or blue azurite—a combination you won’t see with garnets. Finally, while a garnet’s light properties can be measured with standard tools, cuprite’s light-bending power is so high that it goes “off the charts” for most common gem-testing equipment.
Cuprite remains a distinct species in the mineralogical world, primarily defined by its extreme optical and physical constants. Its refractive index, which exceeds that of diamond, and its high copper-driven density make it a subject of significant interest for both gemologists and mineral collectors. While its inherent softness limits its practical application in everyday jewelry, these same constraints highlight its status as a specialized collector’s gem.Whether viewed as a raw mineral specimen or a rare faceted stone, cuprite serves as a clear example of the unique aesthetic results produced by the oxidation of copper deposits. Understanding its diagnostic properties—from its metallic luster to its specific gravity—is essential for accurately identifying and valuing this rare oxide mineral within the broader context of gemology.
