Celsian is a rare and intriguing mineral belonging to the feldspar group, specifically categorized as an aluminosilicate of barium. Its chemical formula is represented as BaAl₂Si₂O₈. While most people are familiar with common feldspars like orthoclase or albite, celsian is unique because barium occupies the large cation sites in the crystal lattice instead of the more typical potassium, sodium, or calcium. Physically, celsian is often colorless, white, or yellow, characterized by a monoclinic crystal system. It possesses a Mohs hardness of 6 to 6.5 and a notably high specific gravity of approximately 3.2 to 3.4, which makes it significantly heavier than its more common cousins due to the presence of the dense barium atom.

Geologic Formation and Occurrence of Celsian
The formation of celsian requires highly specific geochemical environments where barium is abundant and silica is present. It primarily occurs in contact metamorphic rocks and certain hydrothermal deposits, often associated with manganese-rich ore bodies. Celsian typically forms at high temperatures and pressures through the recrystallization of barium-bearing sediments or the alteration of barite (BaSO₄). In the Earth’s crust, celsian rarely exists in a perfectly pure state; it is often found in a solid-solution series with potassium feldspar (orthoclase). The intermediate variety, known as hyalophane, contains both barium and potassium and is more common in nature than the pure barium endmember.

Historical Discovery and Naming
Celsian was first identified and described in 1895 by the Swedish mineralogist Hjalmar Sjögren. The type locality—the place where it was first discovered—is the Jakobsberg Manganese Mine in Filipstad, Sweden, a site famous for its diverse and unusual mineralogy. Sjögren chose to name the mineral in honor of Anders Celsius (1701–1744), the Swedish astronomer renowned for developing the Celsius temperature scale. Beyond its interest to mineral collectors and geologists, celsian has gained modern importance in material science. Synthetic celsian glass-ceramics are highly valued for their low thermal expansion and high-temperature stability, making them ideal for specialized applications like radomes and aircraft engine components.
Crystal Structure of Celsian
Celsian crystallizes in the monoclinic crystal system and belongs to the feldspar group of framework aluminosilicates. Its chemical formula, BaAl₂Si₂O₈, reflects a structure in which barium occupies the large cation sites within the crystal lattice, distinguishing it from more common feldspars that contain potassium, sodium, or calcium. This substitution significantly influences both the geometry and physical properties of the mineral.
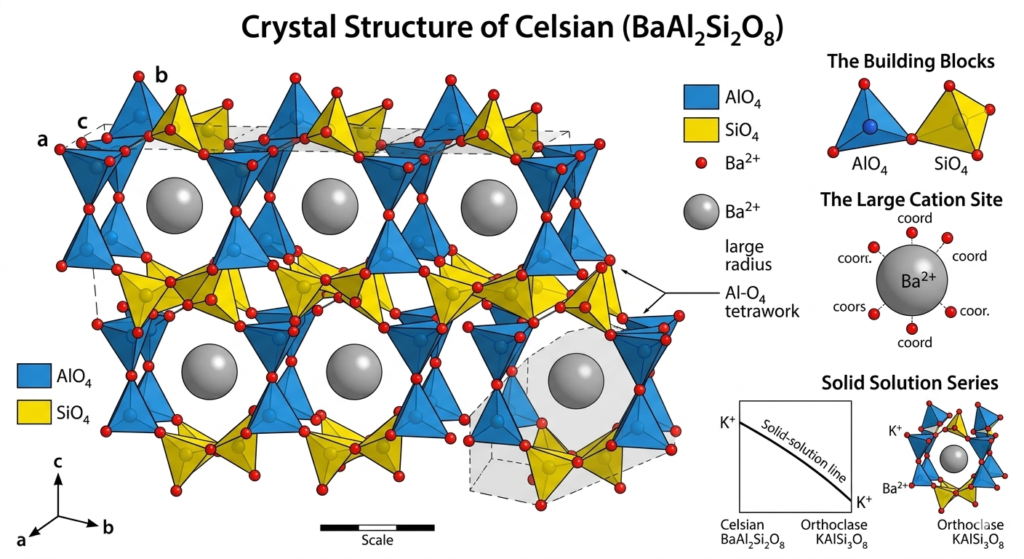
The crystal structure is built from a three-dimensional framework of interconnected aluminum–oxygen and silicon–oxygen tetrahedra. These tetrahedra share oxygen atoms to form a rigid network, with barium ions located in the cavities between them. The relatively large ionic radius and high atomic mass of barium contribute to celsian’s higher density compared to other feldspars and play an important role in stabilizing the structure under specific pressure–temperature conditions.Celsian may also exhibit structural variations related to temperature and composition, particularly within the solid-solution series it forms with potassium feldspar. These variations can affect symmetry and ordering within the lattice, making celsian an important subject in crystallographic and mineralogical studies. Its structure provides insight into how large cations like barium influence the formation and stability of feldspar minerals.
Chemical Composition of Celsian
Celsian is a rare framework silicate mineral that serves as the barium endmember of the feldspar group. Its chemical composition is defined by the formula BaAl₂Si₂O₈. In this structure, the mineral is primarily composed of barium oxide (BaO), aluminum oxide (Al₂O₃), and silicon dioxide (SiO₂). Specifically, a pure specimen typically contains approximately 40.1% BaO, 26.7% Al₂O₃, and 31.4% SiO₂ by weight.

The chemical identity of celsian is characterized by the presence of the large barium divalent cation, which occupies the interstitial sites within the three-dimensional network of aluminum and silicon tetrahedra. Because barium has a significantly larger ionic radius and higher atomic mass than the calcium or potassium found in more common feldspars, celsian exhibits a much higher density and unique crystallographic properties.In natural geological settings, celsian frequently forms a solid-solution series with potassium feldspar (KAlSi₃O₈). This means that barium and potassium cations can substitute for one another within the crystal lattice. When the barium content is intermediate, the mineral is often identified as hyalophane. For a mineral to be strictly classified as celsian, barium must be the dominant large cation. Additionally, minor amounts of other elements such as strontium, iron, or magnesium may occasionally be present as trace impurities, depending on the specific environment of formation, such as manganese-rich metamorphic deposits or hydrothermal veins.
Physical & Optical Properties of Celsian
Celsian exhibits several distinct physical characteristics that separate it from the more common members of the feldspar group. Most notably, its high barium content results in a specific gravity of approximately 3.2 to 3.4. This is significantly higher than that of orthoclase or albite, which typically range from 2.5 to 2.6, making celsian feel noticeably heavier to the touch. It possesses a Mohs hardness of 6 to 6.5, consistent with other feldspars, meaning it can scratch glass but is susceptible to being scratched by quartz. The mineral is typically colorless, white, or pale yellow, with a vitreous to pearly luster on its cleavage surfaces. It demonstrates perfect cleavage in two directions, intersecting at nearly 90 degrees, which is a hallmark of the monoclinic crystal system in which it crystallizes.

Optically, celsian is classified as a biaxial mineral, and it can be either positive or negative depending on its exact composition and the degree of potassium substitution. It has a relatively high refractive index for a feldspar, generally ranging between 1.57 and 1.60. Under a polarizing microscope, celsian shows low birefringence, appearing in shades of first-order gray or white. One of its most identifying optical features is its transparency; while it can be transparent in high-quality crystals, it is more commonly translucent or even opaque in massive forms. Unlike some other feldspars, celsian rarely displays the dramatic play of color known as labradorescence, though it may occasionally show a subtle chatoyancy or pearlescence due to internal structural features.
Identification & How Celsian Differs from Other Feldspars
Identifying celsian can be challenging due to its resemblance to other feldspar minerals, but there are key characteristics that set it apart. The most distinguishing feature of celsian is its barium content, which replaces the more common potassium, sodium, or calcium found in other feldspars. This substitution results in unique physical properties such as a higher specific gravity (3.2–3.4) compared to its feldspar counterparts, making celsian noticeably denser and heavier. It also crystallizes in the monoclinic system, unlike the more common triclinic system of other feldspars like albite or orthoclase.Another identifying feature of celsian is its color and optical properties. While feldspars such as orthoclase or albite are usually colorless, white, or cream, celsian can also appear pale yellow, due to the presence of barium. Its biaxial optical character—a feature common to feldspars—can be observed under polarized light, though its specific refractive indices and birefringence are typically higher than those of other feldspars. Additionally, celsian rarely occurs in its pure form and is often found in a solid-solution series with potassium feldspar, making it important to examine its composition through chemical analysis to ensure correct identification.

Metaphysical & Symbolic Meanings of Celsian
While celsian is primarily valued for its scientific and mineralogical properties, it also holds symbolic and metaphysical significance in various cultures and spiritual practices. As a mineral that contains barium, a dense and heavy element, celsian is often associated with grounding and stability. It is thought to help individuals feel more centered and connected to the Earth’s energy, providing a sense of security during times of emotional or physical imbalance.In metaphysical circles, celsian is believed to enhance mental clarity and insight, aiding in the resolution of mental blocks or confusion. Its high density and solid structure are seen as symbolic of strength, resilience, and endurance, qualities that can be helpful when facing challenging situations or making significant life decisions. Celsian is also thought to promote spiritual growth, offering guidance and protection during meditation or spiritual journeys.
As a symbol of transformation, celsian is often linked to the alteration of energy in both personal and environmental contexts. Its association with high-temperature processes in the Earth’s crust makes it a powerful stone for those seeking to manifest change or renewal in their lives. Whether used for physical healing or emotional well-being, celsian is thought to bring balance, strength, and positive transformation to its wearer or user.
