Millerite is a specialized nickel sulfide mineral with the chemical formula NiS, recognized in the mineralogical community for its exceptional crystal habit and significant geochemical implications. While it serves as a subordinate ore of nickel compared to primary sources like pentlandite, it is highly regarded for its unique occurrence in low-temperature hydrothermal veins and as a secondary alteration product in carbonate-hosted environments. The most defining characteristic of Millerite is its trigonal crystal structure, which almost exclusively manifests as elongated, acicular, or capillary forms. These delicate, hair-like crystals often grow in radiating clusters or intertwined, nest-like masses within rock cavities, displaying a brilliant metallic luster that ranges from pale brass-yellow to a deeper bronze hue upon tarnishing. Beyond its aesthetic appeal to collectors, Millerite provides essential data regarding the geological conditions of its formation, typically developing in environments where nickel-rich fluids interact with sulfur at relatively low temperatures, often in association with minerals such as dolomite, calcite, and other sulfides like chalcopyrite. Geologically, its presence can indicate specific mineralizing processes within serpentinites or as a replacement mineral in nickel-bearing rocks, and with a Mohs hardness of 3 to 3.5 and a high specific gravity of approximately 5.3 to 5.5, its remarkably thin and brittle crystals make finding well-preserved, undamaged specimens a rarity, further cementing its status as a prized highlight in systematic mineralogy databases and specialized geological research.

Formation and Historical Evolution of Millerite
Millerite typically forms through low-temperature hydrothermal processes, often appearing in the cavities, vugs, and veins of sedimentary rocks like limestone and dolomite. It crystallizes when nickel-bearing fluids interact with sulfur at moderate temperatures, allowing the mineral to precipitate slowly into its characteristic needle-like forms. Beyond primary hydrothermal deposition, Millerite is frequently found as a secondary mineral resulting from the alteration of other nickel sulfides or through the serpentinization of ultramafic rocks, where circulating fluids redistribute nickel into rock fractures.
Historically, the mineral was formally described in 1845 by Wilhelm Haidinger, who named it in honor of William Hallowes Miller, the British mineralogist responsible for the Miller Indices used in crystallography. Before this official classification, it was often referred to colloquially as “capillary pyrite” or “hair pyrite” due to its brassy luster and exceptionally thin, thread-like crystals. Notable 19th-century discoveries in regions such as Bohemia and at the Gap Mine in Pennsylvania provided the first significant specimens for study, helping researchers categorize its trigonal symmetry and establish its place within the broader study of sulfide mineralogy.

Varieties and Common Habits of Millerite
Acicular and Capillary Habits
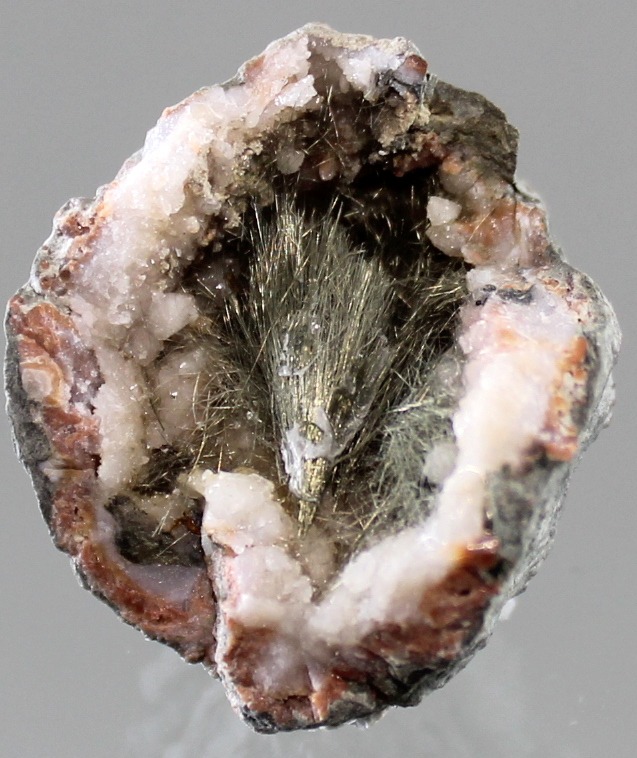
This is the most recognized form of Millerite. It consists of extremely thin, needle-like (acicular) or hair-like (capillary) crystals. These often grow in radiating clusters or intertwined, nest-like masses within rock cavities. Despite their fragility, these crystals maintain a brilliant metallic luster and a pale brass-yellow hue, making them highly sought after by collectors.

Massive and Granular Forms
In some industrial ore settings, Millerite does not form delicate needles but instead appears as dense, massive, or granular aggregates. In this form, it lacks the visual elegance of the capillary variety and is often intermixed with other sulfide minerals. These massive forms are typically identified through chemical assay or microscopic examination rather than visual inspection.

Alteration and Secondary Phases
Millerite frequently occurs as a secondary mineral resulting from the alteration of other nickel-rich sulfides. For instance, in ultramafic rocks, primary pentlandite may transform into Millerite due to late-stage hydrothermal activity. In some cases, Millerite itself may be replaced by other minerals, forming pseudomorphs where the internal composition changes while the original needle-like exterior shape remains.

Distinctive Geological Assemblages
Millerite is often categorized by its host environment, which dictates its physical presentation:
Carbonate-Hosted: Found within geodes in limestone or dolomite, often appearing as pristine, isolated needles alongside calcite or fluorite.
Sulfide Vein-Hosted: Occurs in deep-seated hydrothermal veins associated with chalcopyrite and pyrrhotite, typically found in major nickel mining districts.
Practical Uses and Value of Millerite
Millerite primarily serves as a specialized source of nickel, extracted for its high metal content and processed for use in the production of stainless steel, high-strength alloys, and battery components for the renewable energy sector. Although it is less common than primary ores like pentlandite, its high nickel-to-sulfur ratio makes it a valuable secondary ore in specific geological deposits. Beyond direct extraction, the mineral acts as a strategic indicator in economic geology; its presence in drill cores helps geologists map the chemical evolution of hydrothermal systems and locate broader zones of nickel enrichment. In the scientific community, its distinctive trigonal symmetry and acicular habit provide practical data for researchers studying crystal growth and fluid dynamics. Furthermore, Millerite holds significant value within the specialized mineral specimen market, where well-preserved clusters are traded among museums and private collectors as rare examples of unique sulfide crystallization.
