What Is Diamond?
From a rigorous mineralogical perspective, a diamond is much more than a gemstone; it is the most concentrated and stable allotrope of pure carbon found in nature. What defines a diamond’s extraordinary character is its diamond cubic crystal structure, where each carbon atom is locked into a rigid, three-dimensional tetrahedral lattice through intense covalent bonding. This unique atomic arrangement is the scientific secret behind its legendary status as the hardest natural substance on Earth, earning a definitive 10 on the Mohs scale. Unlike most other minerals that are chemical compounds of multiple elements, the diamond’s elemental purity and the extreme density of its atoms allow it to resist scratching and chemical erosion better than any other material, making it not only a symbol of eternal love for April birthdays but also an indispensable tool in high-tech industrial and scientific applications.

History of Diamonds: From Ancient India to Modern Luxury
The history of diamonds begins in the riverbeds of ancient India, where the first recorded stones were discovered over three thousand years ago. Initially valued for their extreme hardness and ability to refract light, these early diamonds were used as religious icons and engraving tools rather than personal adornment. By the fourth century BCE, diamonds had become a prized commodity, traded along the Silk Road reaching as far as China and the Mediterranean. For centuries, India remained the world’s only known source of diamonds, producing legendary stones such as the Koh-i-Noor, which came from the prolific Golconda mines.

During the Middle Ages and the Renaissance, diamonds began to migrate into European royal treasuries. However, it was not until the fourteenth century that the art of diamond cutting began to evolve, transforming dull, octahedral crystals into faceted gemstones that could finally showcase their internal brilliance. The discovery of diamonds in Brazil during the early eighteenth century briefly shifted the global supply chain after India’s mines began to deplete. Yet, the modern diamond industry as it is recognized today was truly born in the late 1860s with the discovery of massive primary deposits in Kimberley, South Africa. This find marked the transition from diamonds being an ultra-rare gemstone reserved for the highest nobility to a cornerstone of the global luxury market.
In the twentieth century, the narrative of the diamond was further reshaped by sophisticated marketing and industrial standardization. The introduction of the four Cs—carat, color, clarity, and cut—by the Gemological Institute of America provided a universal language for evaluating diamond quality, bringing transparency to the trade. Today, the industry continues to evolve through the integration of ethical sourcing protocols and the emergence of laboratory-grown alternatives. From their origins as sacred talismans in the Vedic period to their current status as the ultimate symbol of commitment and craftsmanship, diamonds remain one of the most enduring intersections of geological wonder and human cultural history.
How Are Diamonds Formed in Nature?
Natural diamonds are formed deep within the Earth’s mantle, approximately 150 to 250 kilometers below the surface, where pure carbon is subjected to pressures of up to 60,000 atmospheres and temperatures exceeding 1,100°C. Under these extreme conditions, carbon atoms are forced into a rigid, three-dimensional tetrahedral lattice known as a diamond cubic crystal structure, resulting in the hardest natural substance known to science. These crystals remain in the mantle for millions or even billions of years until they are transported to the surface by rare, deep-seated volcanic eruptions through kimberlite or lamproite pipes. This violent ascent occurs at high speeds, cooling the magma quickly enough to prevent the diamonds from turning into graphite, ultimately preserving their unique atomic bond and unmatched brilliance.
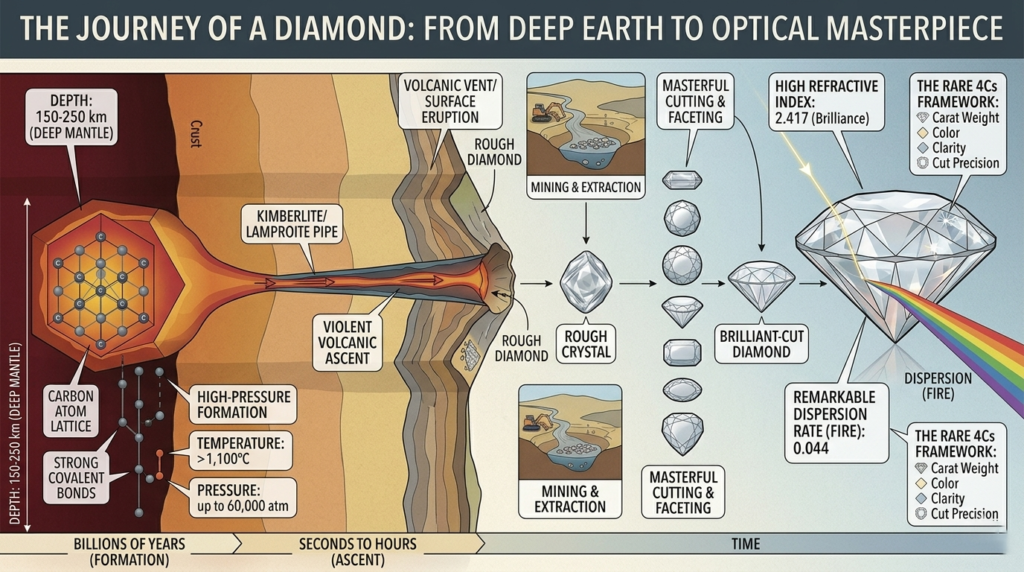
Why Diamonds Are the Hardest Natural Material
The unmatched hardness of a diamond is rooted in its unique atomic architecture and the specific nature of its chemical bonding. As a pure form of carbon, each atom within a diamond is linked to four neighboring carbon atoms through exceptionally strong covalent bonds, forming a rigid, three-dimensional tetrahedral lattice. This crystalline structure ensures that the atoms are packed incredibly densely, leaving no planes of weakness for the material to be easily displaced or scratched.On the Mohs scale of mineral hardness, diamonds occupy the definitive position of 10, meaning they can only be scratched by another diamond. This extreme durability is not merely a result of the element itself—as seen in graphite, which is also pure carbon but remains one of the softest minerals—but rather the way the atoms are organized under the immense pressure of the Earth’s mantle. This combination of elemental purity and a flawless, interconnected geometry makes the diamond the ultimate natural material for both high-end jewelry and demanding industrial cutting and grinding applications.
Diamond Crystal Structure Explained
The extraordinary physical properties of a diamond, from its extreme hardness to its high thermal conductivity, are the direct result of its sophisticated atomic arrangement. At its core, a diamond is a crystalline form of pure carbon where every atom is locked into a rigid, three-dimensional network known as a diamond cubic crystal structure.
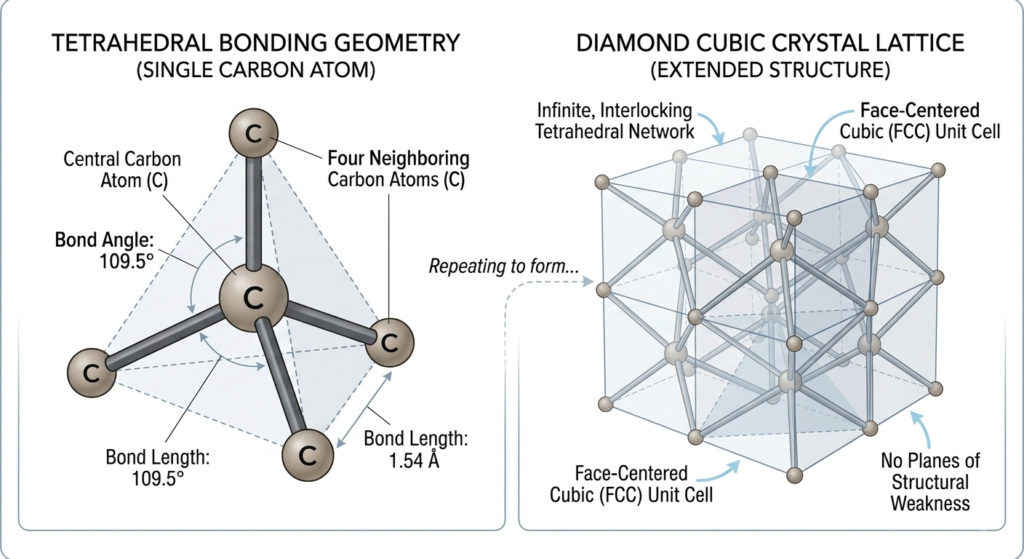
In this configuration, each carbon atom is covalently bonded to four neighboring carbon atoms, located at the corners of a regular tetrahedron. These covalent bonds are among the strongest chemical bonds in nature, requiring immense energy to break. Unlike graphite, where carbon atoms are arranged in loosely bonded sheets that can slide past one another, the atoms in a diamond are interconnected in all directions. This uniform, dense packing ensures that there are no natural planes of structural weakness, which is why a diamond can only be scratched by another diamond.The symmetry of this tetrahedral lattice also plays a critical role in the gemstone’s optical performance. Because the atoms are arranged with such high precision, light entering the crystal is reflected and refracted with minimal interference, allowing for the high refractive index and dispersion that define a diamond’s brilliance. From a mineralogical perspective, this structure represents the most stable and compact arrangement of carbon atoms possible under high-pressure conditions, serving as a perfect example of how atomic-level geometry dictates macro-level physical excellence.
Understanding the 4Cs of Diamond Quality
The 4Cs—Carat, Color, Clarity, and Cut—represent the universal standard for defining the quality and value of a diamond. Established by the Gemological Institute of America (GIA) in the mid-twentieth century, this grading system replaced a chaotic marketplace of conflicting terms with a consistent, scientific language. Together, these four characteristics determine the rarity of a gemstone and dictate its global market price.
Cut
The cut of a diamond is often considered the most critical of the 4Cs because it directly impacts the stone’s ability to reflect light. A well-proportioned cut allows light to enter through the table, bounce off the internal facets, and return to the eye as fire and brilliance. If a diamond is cut too shallow or too deep, light leaks out of the sides or bottom, resulting in a dull or “dark” appearance. The cut grade specifically evaluates the craftsmanship of the faceting rather than the shape of the diamond itself.
Evaluating Diamond Cut Grade
Select a grade below to visualize light performance based on GIA standards.
Color
The color of a diamond is one of the most significant factors in determining its rarity and market value. According to the international standards established by the Gemological Institute of America (GIA), white diamonds are graded on a scale from D (colorless) to Z (light yellow or brown). This grading process is conducted under controlled lighting conditions by comparing each stone against a set of master stones. As a diamond moves down the scale from D toward Z, the presence of subtle yellow or brown tints becomes more apparent, which typically leads to a decrease in the stone’s price per carat.While the differences between adjacent grades, such as E and F, are virtually invisible to the untrained eye, they represent distinct levels of chemical purity. Diamonds in the D-E-F range are classified as colorless and are prized for their icy brilliance. Stones in the G-H-I-J range are near-colorless and appear white when set in jewelry, offering an excellent balance of visual appeal and value. Beyond the K grade, the warmth of the stone becomes noticeable, which some collectors appreciate for its vintage character, though these stones are more abundant in nature compared to their colorless counterparts.

Clarity
Because diamonds are formed under extreme pressure deep within the Earth, most contain unique birthmarks known as inclusions (internal) or blemishes (external). Clarity is the measure of the number, size, and placement of these characteristics. The scale ranges from Flawless, indicating no visible inclusions under 10x magnification, to Included, where characteristics may be visible to the naked eye. Most diamonds fall into the VS (Very Slightly Included) or SI (Slightly Included) categories, where inclusions do not affect the structural integrity or overall beauty.
Microscopic Clarity Grading
Simulated 10x Magnification View
Carat
Carat refers specifically to the weight of the diamond, not its physical size. One carat is defined as exactly 200 milligrams. Because large, high-quality diamonds are found much less frequently in nature than smaller ones, the price of a diamond increases exponentially as the carat weight rises. This means a single two-carat diamond will cost significantly more than two one-carat diamonds of equal quality, reflecting the extreme rarity of larger crystals.
The Influence of Diamond Shape in Jewelry Design and Application
In the realm of high jewelry, the shape of a diamond is the foundational element that defines a piece’s character, silhouette, and overall aesthetic narrative. While the cut grade measures the technical precision of facets and light return, the shape represents the artistic geometry that captures a wearer’s personal style, making the selection process a critical bridge between gemological science and wearable art. The Round Brilliant cut remains the most iconic and mathematically perfected shape, engineered with 57 or 58 facets to achieve maximum brilliance and fire while effectively masking minor internal inclusions. For designs prioritizing sophisticated clarity, step-cut diamonds like the Emerald and Asscher shapes offer a “hall-of-mirrors” effect through long, rectangular facets that radiate understated luxury. Fancy shapes, including the modern Princess cut and elongated options like the Oval, Pear, and Marquise, allow for significant creative expression and can strategically optimize the perceived size of a stone relative to its carat weight. Specialty geometries such as the Heart and Cushion cuts cater to romantic and vintage-inspired niches, ensuring that every application of a diamond shape is a deliberate balance of light performance, durability, and visual impact.
Explore Diamond Shapes

The most popular shape, engineered for unmatched fire and brilliance.
 Round
Round
 Oval
Oval
 Emerald
Emerald
 Cushion
Cushion
 Pear
Pear
 Radiant
Radiant
 Princess
Princess
 Marquise
Marquise
 Asscher
Asscher
 Heart
Heart
Natural vs Lab-Grown Diamonds
Laboratory-grown diamonds are produced through advanced technological processes that replicate the extreme conditions found deep within the Earth’s mantle. There are two primary methods used to create these stones: High Pressure High Temperature (HPHT) and Chemical Vapor Deposition (CVD). In the HPHT method, a small diamond seed is placed in a carbon source and subjected to intense pressure and heat—reaching over 1,400°C—using heavy machinery such as cubic or belt presses to mimic natural geological forces. Alternatively, the CVD process involves placing a diamond seed in a vacuum chamber filled with carbon-rich gases, which are then ionized into plasma; the carbon atoms subsequently break down and precipitate onto the seed, growing the crystal layer by layer. Because both methods result in a material with the identical chemical, physical, and optical properties as natural diamonds, synthetic stones are considered real diamonds rather than imitations.
Natural vs Lab-Grown Diamonds: A Comprehensive Comparison
| Dimension | Natural Diamonds | Lab-Grown Diamonds |
|---|---|---|
| Geological Origin | Formed approximately 150 to 250 kilometers deep within the Earth’s mantle under extreme pressure and heat over billions of years. | Produced in controlled laboratory environments using HPHT or CVD technology to replicate natural conditions within weeks. |
| Chemical Structure | Composed of pure carbon arranged in a tetrahedral crystal lattice; often contains trace amounts of nitrogen or other Earth minerals. | Composed of pure carbon with an identical tetrahedral crystal lattice; typically exhibits higher elemental purity due to controlled growth. |
| Physical Durability | The hardest natural material known to science, scoring a perfect 10 on the Mohs scale with an adamantine luster. | Possesses the exact same physical integrity as natural stones, scoring a 10 on the Mohs scale with identical scratch resistance. |
| Optical Brilliance | Features a refractive index of 2.417 and a dispersion rate of 0.044, creating the characteristic fire and scintillation. | Displays the same refractive index of 2.417 and dispersion of 0.044, resulting in visual properties indistinguishable from mined stones. |
| Market Scarcity | A finite, non-renewable natural resource with a supply limited by geological discovery and mining extraction. | A manufactured product with a scalable supply chain; production is limited only by technological capacity and laboratory runtime. |
| Industry Value | Command higher market prices and maintain significant resale value as a luxury asset and collectible mineral. | Offered at a lower price point, typically 30 to 70 percent less than natural stones, focused on accessibility and consumer choice. |
| Authority Grading | Certified by the GIA or IGI as a natural diamond of volcanic origin, verified through spectroscopic analysis of nitrogen levels. | Certified by the GIA or IGI as a laboratory-grown diamond, often featuring a microscopic laser inscription to ensure transparency. |
According to the Gemological Institute of America (GIA) and the Federal Trade Commission (FTC), lab-grown diamonds are chemically, physically, and optically identical to natural diamonds. While they share the same crystal structure and brilliance, their origins and market positions represent two distinct categories of gemstones. Natural diamonds are rare geological artifacts formed 150 to 250 kilometers deep within the Earth’s mantle over one to three billion years. As noted by the Smithsonian Institution, these stones are brought to the surface by rare volcanic pipes, making them a finite natural resource. Conversely, lab-grown diamonds are produced in controlled environments using High Pressure High Temperature (HPHT) or Chemical Vapor Deposition (CVD). These methods replicate the intense heat and pressure of the Earth, but complete the growth cycle in weeks rather than eons.The primary distinction between the two lies in their rarity and long-term value. Reports from major industry analysts like Bain & Company highlight that natural diamonds derive their value from their scarcity and the complex global supply chain required to extract them. This inherent rarity allows natural diamonds to maintain a higher resale value and status as a luxury asset. Lab-grown diamonds, being products of scalable manufacturing, have seen a steady decline in production costs as technology advances. This makes them an excellent choice for consumers prioritizing size and clarity at a more accessible price point, though they typically do not hold the same secondary market value as Earth-mined stones over the long term.
To the naked eye, even a master gemologist cannot distinguish a lab-grown diamond from a natural one. Scientific identification requires specialized spectroscopic equipment used by major laboratories like the GIA or the International Gemological Institute (IGI). These professional tools detect minute growth patterns and trace elements, such as specific nitrogen levels in natural stones or metallic flux remnants in HPHT diamonds. To ensure full consumer transparency, all reputable lab-grown diamonds are laser-inscribed with a unique report number and the phrase Laboratory-Grown, accompanied by a formal grading report from an authoritative body that explicitly states the origin of the gemstone.

Scientific Methods for Diamond Identification
Distinguishing between natural and laboratory-grown structures through advanced gemology.
How to Clean a Diamond Safely
Maintaining the breathtaking sparkle of your diamond requires regular, gentle cleaning to remove the buildup of oils and debris that naturally accumulate through daily wear. To clean your diamond safely at home, soak the jewelry in a solution of warm water and a few drops of mild, fragrance-free dish soap for approximately 20 to 30 minutes. Use a new, soft-bristled toothbrush to gently scrub the facets and reach the hard-to-access areas underneath the setting, as this is where most grime collects. After scrubbing, rinse the piece thoroughly under warm running water—ensuring the drain is plugged—and pat it dry with a lint-free microfiber cloth. Avoid using harsh chemicals like bleach or abrasive cleaners, as these can damage the metal setting or diminish the natural brilliance of the stone. For more detailed information, please visit our gemstone cleaning guide.
